AUGUST 23, 2020
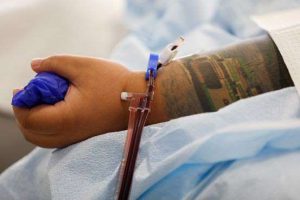
The Food and Drug Administration authorized use of convalescent plasma, the antibody-rich blood component taken from recovered Covid-19 patients, for the treatment of serious coronavirus cases.
The agency’s action Sunday, called an emergency-use authorization, permits use of the treatment on hospitalized Covid-19 patients. The Wall Street Journal reported last month the move was coming.
For Covid-19 patients and the doctors who treat them, the designation opens up the possibility for faster and easier access to a promising treatment, while studies and clinical trials continue to explore who is helped and how much.
Doctors have been looking for validated coronavirus treatments. Until now, only one other drug, remdesivir from Gilead Sciences Inc., has the FDA’s emergency-use OK.
Certain seriously ill Covid-19 patients treated with plasma containing high levels of antibodies appeared to benefit more than those who received low levels of antibodies, said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research.
Hospitalized patients who received the plasma within three days of diagnosis, are under the age of 80 and not on mechanical ventilation, benefited the most, with a 35% improvement in survival 30 days after receiving the transfusion compared with patients who got plasma with low antibody levels, according to Dr. Marks.
“We’re confident that convalescent plasma is safe to use in this setting,” Dr. Marks added.
President Trump, at a White House news conference Sunday, described the FDA’s move as a breakthrough that makes the product far more widely available than it has been. He also took credit for the timing, saying his administration removed barriers delaying action.
FDA Commissioner Stephen Hahn said the authorization falls short of a full approval and the agency will evaluate more evidence.
Convalescent plasma has been seen as a way to help people fight the disease and a bridge while other treatments are under development. The emergency-use authorization doesn’t alleviate the need for a vaccine or for therapies known as monoclonal antibodies that could stave off infection or at least reduce the seriousness of one.
Emergency-use authorization waives some regulatory requirements involved in using products during public health emergencies that aren’t yet FDA-approved.
Mr. Trump, who has spread messages attacking scientists including National Institute of Allergy and Infectious Diseases Director Anthony Fauci, criticized the FDA in a tweet yesterday, saying the agency was impeding efforts to roll out coronavirus vaccines and drugs for political reasons.
Mr. Hahn, the FDA commissioner, was nominated by Mr. Trump. The vaccines and drugs furthest along in development are moving at a remarkable speed, industry and government officials say. Several are now in late-stage testing, which the agency typically requires to assure they work safely.
White House chief of staff Mark Meadows suggested on the show “Fox News Sunday” the FDA could have moved faster to issue the emergency-use authorization.
Democrats criticized the president and his administration for politicizing the FDA. “The FDA must approve drugs or vaccines based on their safety and effectiveness—NOT political pressure from the White House,” House Speaker Nancy Pelosi said in a tweet on Saturday.
The agency had been moving toward greenlighting convalescent plasma for weeks, according to people familiar with the matter. At the request of the National Institutes of Health, last week it examined new data from the Mayo Clinic and colleagues who have been evaluating the treatment in thousands of Covid-19 patients, according to a person familiar with the matter.
The FDA’s action is based primarily on analyses of recent data from a large expanded-access program sponsored by the agency and led by the Mayo Clinic in Rochester, Minn. More than 72,000 Covid-19 patients have been transfused to date through the program, though the FDA relied on data from a fewer number of patients for its analysis.
The Mayo Clinic researchers reported, in a paper posted on a public server earlier this month, that patients treated with plasma with high levels of antibodies within three days of diagnosis had a mortality rate of 8.7% at seven days after the transfusion, compared with a mortality rate of 11.9% for patients who got plasma with low levels of antibodies at four days or more after diagnosis.
The analysis has limitations. It hasn’t been published in a journal or subjected to peer review. Also, expanded-access programs aren’t as rigorous as a clinical trial, which compares how subjects who got the study drug did at random with subjects who received a placebo or the standard of care.
Given the shortcomings, Mayo Clinic researchers can’t definitively say whether plasma improves outcomes because every patient in the study receives it. Rather, they have reported signs the treatment might be working in certain patients.
Other studies also have indicated that patients receiving high-antibody plasma early in the illness appear to benefit.
Researchers involved in a continuing study of more than 300 Covid-19 patients treated with convalescent plasma at Houston Methodist published a study earlier this month in the American Journal of Pathology found those who received high-antibody plasma within 72 hours after hospitalization were more likely to survive than similar patients not treated with plasma.
FDA’s Dr. Marks said the data used for the emergency authorization are preliminary and the agency will consider updating the authorization as more information is available.
Courtesy/Source: WSJ