APRIL 20, 2020
COVID-19 testing
After weeks of sheltering in place, Americans are asking how soon we can return to a more normal life outside our homes. Much of the answer might be in a test.
The first phase of testing has been about determining who has COVID-19. The next phase will be about who had it – or may still be fighting it.
Instead of looking in our throats for the coronavirus itself, health care workers will look for signs in our blood that we developed antibodies to fight the virus.
The results could tell us many things.
Infection rates. Researchers say many people have been asymptomatic – or didn’t know they were inflected. Widespread testing would provide insights into how prevalent the most serious cases have been.
Herd immunity. If a large percentage of people in a city or state have those antibodies, researchers might determine there is enough herd immunity to protect those who haven’t contracted the virus.
Marc Lipsitch, a professor of epidemiology at Harvard and an expert in public health interventions, told the USA TODAY Editorial Board on April 8 that a significant portion of the population must be immune to the coronavirus before social distancing restrictions can be lifted.
Unproven tests. Inaccurate results: Public health labs worry ‘bad data’ could taint U.S. recovery from coronavirus crisis.
What is an antibody?
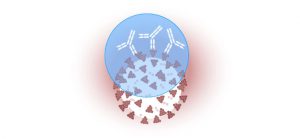
Antibodies are the body’s way of remembering how it responded to an infection so it can attack again if exposed to the same pathogen. People with antibodies in their blood have immune cells available to fight the virus, which lowers the risk of reinfection.
Antibodies are Y-shaped proteins produced by white blood cells to help stop a virus from intruding. Antibody tests look for two antibodies in the blood, immunoglobulin M (IgM) and immunoglobulin G (IgG).
IgM and IgG antibodies fight all kinds of infections. The blood tests for COVID-19 look for antibodies specific to this coronavirus, which shows whether the body is producing antibodies to it or to others such as the seasonal flu.
How tests find coronavirus antibodies
To create an antibody test, researchers isolate specific parts of the genetic material from a virus that correspond to the virus’ outermost vulnerable layer – the place where antibodies attach themselves.
Once researchers isolate specific parts of the virus’ genetic code, or RNA, they inject it into other types of mammal cells to make those cells grow with the same structure, or outermost shell, as the novel coronavirus.
“As they grow, they’ll start to produce the spike protein,” University of Arizona immunologist Deepta Bhattacharya said. “You can basically trick the cells into making what you want.”
How to test for coronavirus antigens
The entire virus structure is known as an antigen – a foreign substance that engages the immune system. An effective lab-grown spike protein would have the same shape as the novel coronavirus.
Health care workers take a sample of a patient’s blood and isolate the parts of the blood that contain the antibodies into a serum. If a patient has coronavirus antibodies, they would be found in the blood, along with the other antibodies.
Other tests can involve just a simple finger prick to draw a blood sample, which is collected with a plastic straw and deposited in a small cartridge along with a special solution of liquids that will cause a reaction. Ten minutes later, the test promises to tell you whether you’ve had COVID-19.
These tests are one of many antibody tests the FDA approved under its Emergency Use Authorization, which means they haven’t been thoroughly reviewed by the agency and are not guaranteed to be accurate.
In a clinical evaluation, Zhejiang Orient Gene Biotech reported the finger-prick tests to be 61.8% to 94.4% sensitive, depending on the type of antibody that shows up in the test results, which means that the tests are reported to give accurate results 61.8% to 94.4% of the time.
The challenge of creating a coronavirus blood test
Dr. Raed Dweik, chairman of the Respiratory Institute at the Cleveland Clinic, said some of the tests he has seen aren’t accurate enough to determine whether someone is truly immune to the coronavirus.
That’s because antibodies for the coronavirus that causes COVID-19 look very similar to other coronavirus antibodies that cause illnesses, such as the common cold.
Tests could mistakenly identify antibodies as being for the coronavirus that causes COVID-19, producing a false positive. Dweik said it will take more time to develop a test that can accurately detect the right antibodies.
Why antibody testing is key to reopening America
Dr. Neeraj Sood, professor and vice dean of research and faculty at the University of Southern California Sol Price School of Public Policy, leads a study in conjunction with the Los Angeles County Department of Public Health using antibody tests to determine how widely it has spread and how deadly it is.
As of Sunday, there have been more than 600 deaths related to COVID-19 in the county, according to the health department. If, based on the antibody study, researchers determine that about 5,000 people had been infected, it would be considered a deadly disease. If more than 2 million people had been infected, it wouldn’t be considered that dangerous.
“If we find out COVID is far less deadly than the flu, we can open up the economy. You don’t need to hit herd immunity to open it up,” Sood said. “But if you find out that COVID is 10 times deadlier than the flu, then you have to keep it closed.”
Courtesy/Source: USA Today