DECEMBER 7, 2020
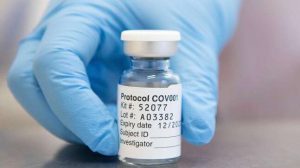
Britain will begin roll-out of the coronavirus vaccine developed by Pfizer and BioNTech on Tuesday to start vaccinating its general population against the novel coronavirus. Just a day before Pfizer’s coronavirus vaccine roll-out in the UK, a senior official said India is accelerating its review of coronavirus vaccines developed by Pfizer Inc and AstraZeneca to authorise them for emergency use.
The government is pinning its hopes for mass supply on the Serum Institute of India, the world’s largest vaccine producer by volume, which lodged the first formal application for emergency-use approval of AstraZeneca’s coronavirus vaccine shot on Monday.
Recently, Pfizer had applied for emergency-use approval for its coronavirus vaccine in India over the weekend. Serum Institue, too, has applied for the emergency approval of the vaccine. On Monday, Bharat Biotech filed an application on Monday seeking emergency-use authorization for its coronavirus vaccine.
The development comes as PM Narendra Modi recently said that a coronavirus vaccine will be available in India in a few weeks. PM Modi said India will begin its coronavirus vaccination programme as soon as the country gets a nod from the scientists.
BRITAIN VACCINE ROLL-OUT
Britain is the worst-hit European country from coronavirus, with over 61,000 deaths, but Prime Minister Boris Johnson hopes to turn the tide against the disease by rolling out the Pfizer/BioNTech vaccine before the United States or European Union.
Britain approved the coronavirus vaccine for emergency use less than a week ago, and is rolling it out through its National Health Service (NHS).
In total Britain has ordered 40 million doses of the Pfizer/BioNTech shot. As each person requires two doses, that is enough to vaccinate 20 million people in the country of 67 million. About 800,000 doses are expected to be available within the first week, with care home residents and carers, the over 80s and some health service workers the top priority to get them.
Courtesy/Source: India Today