SEPTEMBER 23, 2020
- Medicare Part B, which generally covers approved vaccines deemed necessary, does not extend that coverage to those approved under emergency-use authorization, which is expected for an early Covid-19 vaccine.
- It’s uncertain when a legislative fix could come or if there are regulatory maneuvers that could provide a work-around instead.
- A statement from the Centers for Medicare and Medicaid Services said the agency is “committed to modernizing the Medicare program and delivering for our nation’s seniors to ensure access to a vaccine with no cost sharing.”
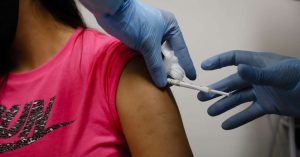
A health worker injects a person during clinical trials for a Covid-19 vaccine at Research Centers of America in Hollywood, Florida.
If you’re on Medicare and hope to get a coronavirus vaccine as soon as it’s available, it may take an act of Congress for it to be free.
The CARES Act, signed into law in March, has no cost-sharing for Medicare beneficiaries for a Covid-19 vaccine once it’s available. However, due to existing Medicare rules, the legislation would apply only to a vaccine that undergoes the standard approval process for new biologics — not one authorized under emergency use, which is how the Trump administration anticipates making a vaccine available quickly.
“I think officials are realizing they don’t have a good solution besides going back to Congress and getting the statutory language changed,” said Juliette Cubanski, deputy director for the Kaiser Family Foundation’s program on Medicare policy.
It’s uncertain when a legislative fix would be proposed, or if there are regulatory maneuvers that could provide a work-around instead.
“This administration is committed to modernizing the Medicare program and delivering for our nation’s seniors to ensure access to a vaccine with no cost-sharing,” according to a statement provided to CNBC from the Centers for Medicare and Medicaid in response to questions about when and how the agency planned to resolve the issue.
The glitch was first reported by The Wall Street Journal.
As a handful of drug manufacturers work on a vaccine as part of the Trump administration’s push for quick availability during this public-health crisis, an emergency-use authorization would fall short of the more rigorous approval process typically required by the Food and Drug Administration. Going that route falls outside the norm when it comes to vaccines or prescription drugs, Cubanski said.
“But there’s clearly a lot of scientific and medical firepower being thrown at the search for a Covid vaccine,” Cubanski added.
The intent by the Centers for Disease Control and Prevention would be to get the limited quantities first available to those most at risk for contracting Covid-19 or dying from complications caused by the virus.
That generally would include Medicare beneficiaries, the majority of whom are age 65 or older — an age group identified as high-risk.
There are about 62.7 million individuals enrolled in Medicare, although not all are enrolled in Part B, which typically covers vaccines. (Some beneficiaries with health coverage elsewhere delay signing up for Part B and instead only get Part A — hospital coverage — because they usually pay no premiums).
Although President Trump has said a vaccine could be ready for emergency use before the end of October — a claim contradicted at times by others in his administration — the FDA is expected to soon release strict guidelines for the accelerated process that essentially would mean emergency approval wouldn’t come until at least November if not later.
Meanwhile, pricing for a vaccine has varied among drug manufacturers, and it remains uncertain which will get approval first. Moderna, one of the pharmaceutical companies working on a vaccine, said in August that it’s charging $32 to $37 per vaccine. In contrast, Pfizer and German biotech BioNTech agreed to $19.50 in a deal with the U.S. government.
However, even as the U.S. races to get a vaccine to the public, new CNBC/Change Research polls show that most likely voters (61%) worry Trump is pushing a vaccine’s release too quickly. Additionally, less than half (42%) said they will definitely or probably get the shot when it first becomes available.
Courtesy/Source: CNBC